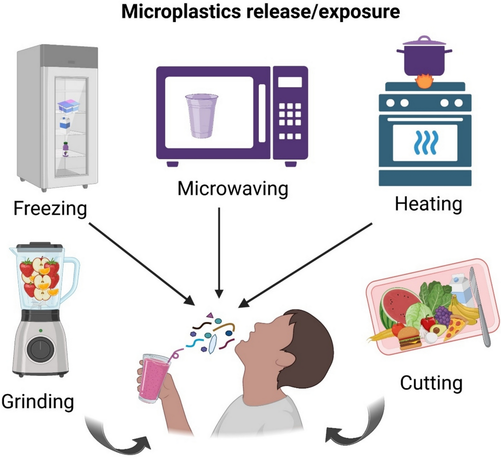
i can imagine toxic impurities being a problem, especially if you get your solvents from the hardware store or cleaning supplies shop; but you can get them in food-safe/pharmaceutical-grade varieties too
i can imagine acetone destroying PTFE coatings, since PTFE itself is a polymer, but i don't have any PTFE cookware except my airfryer and rice cooker
what about other materials? stainless steel shouldn't be a problem. neither should glass, which includes emamel coatings. raw ceramic and cast iron might be a problem since they're porous, but then again, these solvents evaporate pretty quickly.