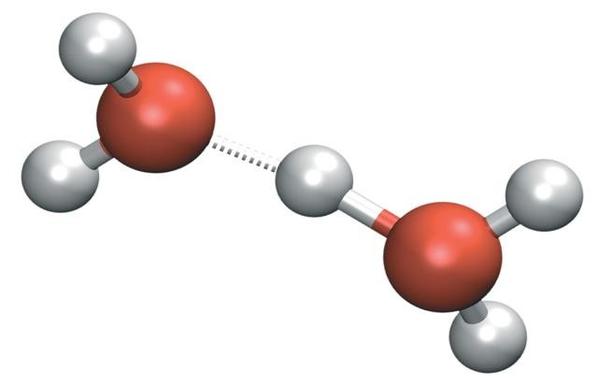
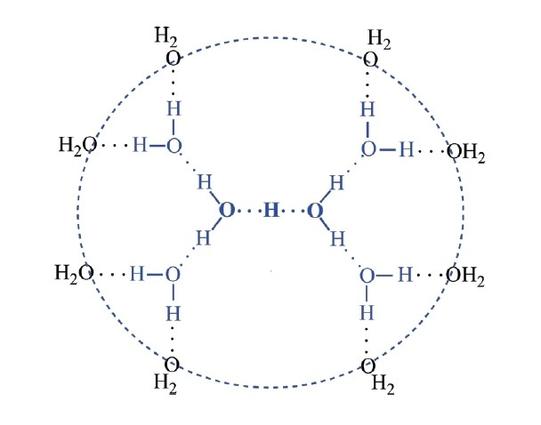
If you could watch an individual water molecule, about once in 10 hours you'd see it do this!
As it bounces around, every so often it hits another water molecule hard enough enough for one to steal a hydrogen nucleus - that is, a proton - from the other!
The water molecule with the missing proton is called a hydroxide ion, OH⁻. The one with an extra proton is called a hydronium ion, H₃O⁺.
This process is called the 'autoionization' of water. Thanks to this, roughly one in ten million molecules in a glass of water are actually OH⁻ or H₃O⁺, not the H₂O you expect.
And this gives a cool way for protons to move through water. Let's watch it!
(1/n)