The complex structure of piperine with the PPARγ ligand-binding domain provides a greater understanding of the way in which piperine modulates PPARγ function and its potential as a candidate for treatment of metabolic disorders #PPARgamma #Piperine https://doi.org/10.1107/S2053230X25002377
Molecular interactions between piperine and peroxisome proliferator-activated receptor gamma ligand-binding domain revealed using co-crystallization studies
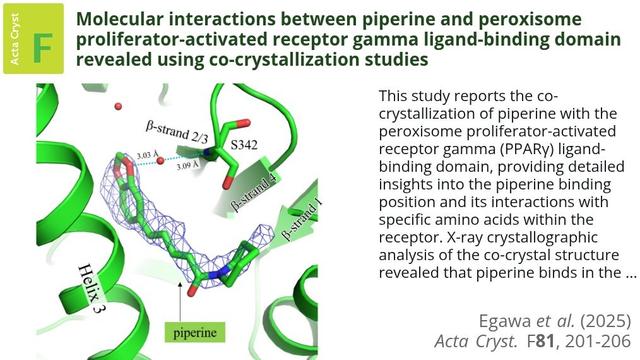
This study reports the co-crystallization of piperine with the peroxisome proliferator-activated receptor gamma (PPARγ) ligand-binding domain, providing detailed insights into the piperine binding position and its interactions with specific amino acids within the receptor. X-ray crystallographic analysis of the co-crystal structure revealed that piperine binds in the ligand-binding pocket of PPARγ via hydrogen-bonding and hydrophobic interactions, suggesting that it plays a role as a partial agonist or antagonist and thereby holds promise as a natural alternative to synthetic PPARγ modulators.