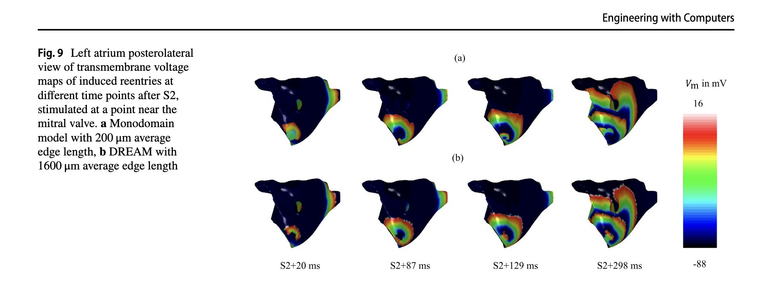
Our use case partner Axel Loewe has published "A cyclical fast iterative method for simulating reentries in cardiac electrophysiology using an eikonal-based model": The DREAM model, which incorporates conduction velocity restitution and an approximated reaction-diffusion component, significantly accelerates the simulation of complex cardiac arrhythmias compared to traditional models.
https://zenodo.org/records/14906903
#openCARP #CardiacModeling #EikonalModel #digitalization #ditrare @fiz_karlsruhe
A cyclical fast iterative method for simulating reentries in cardiac electrophysiology using an eikonal-based model
Computer models for simulating cardiac electrophysiology are valuable tools for research and clinical applications. Traditional reaction–diffusion (RD) models used for these purposes are computationally expensive. While eikonal models offer a faster alternative, they are not well-suited to study cardiac arrhythmias driven by reentrant activity. The present work extends the diffusion–reaction eikonal alternant model (DREAM), incorporating conduction velocity (CV) restitution for simulating complex cardiac arrhythmias. The DREAM modifies the fast iterative method to model cyclical behavior, dynamic boundary conditions, and frequency-dependent anisotropic CV. Additionally, the model alternates with an approximated RD model, using a detailed ionic model for the reaction term and a triple-Gaussian to approximate the diffusion term. The DREAM and monodomain models were compared, simulating reentries in 2D manifolds with different resolutions. The DREAM produced similar results across all resolutions, while experiments with the monodomain model failed at lower resolutions. CV restitution curves obtained using the DREAM closely approximated those produced by the monodomain simulations. Reentry in 2D sheets yielded similar results in vulnerable window and mean reentry duration for low CV in both models. In the left atrium, most inducing points identified by the DREAM were also present in the high-resolution monodomain model. DREAM’s reentry simulations on meshes with an average edge length of 1600µm were 87x faster than monodomain simulations at 20µm . This work establishes the mathematical foundation for using the accelerated DREAM simulation method for cardiac electrophysiology. Cardiac research applications are enabled by a publicly available implementation in the openCARP simulator.