https://doi.org/doi:10.1186/s12929-026-01244-z
https://pubmed.ncbi.nlm.nih.gov/42083040/
#Microtubule
Tubulin glutamylation: a key regulator of flagella, cilia, centrosomes, and disease pathways - Journal of Biomedical Science
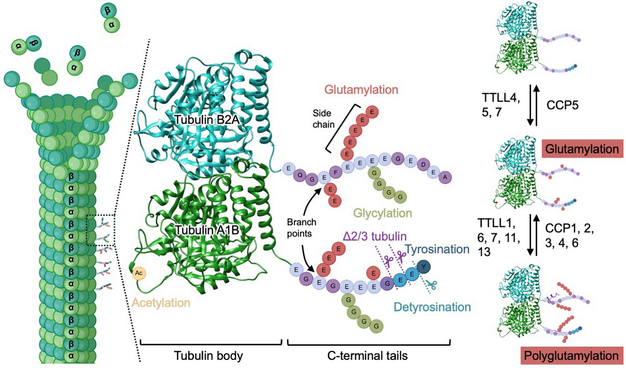
Tubulin glutamylation is an essential post-translational modification that expands the functional diversity of microtubules in many cellular structures, including flagella, motile cilia, primary cilia, centrosomes, and neurons. This modification adds variable lengths of glutamate side chains to the C-terminal tails of tubulin, creating a finely tuned biochemical signal that regulates microtubule stability, motor protein movement, the activity of severing enzymes, and the recruitment of key signaling molecules. Growing evidence shows that glutamylation is not uniformly distributed but instead forms distinct spatial patterns along microtubule arrays, particularly within the axonemes of flagella and cilia, centriolar triplets, and long-lived neuronal microtubules. These patterns are established by tubulin ligase–like enzymes that add glutamates and by carboxypeptidases that remove them, together shaping a dynamic “tubulin code.” In motile cilia and flagella, glutamylation fine-tunes dynein-driven force generation and the coordination of axonemal bending. Disruption of this modification impairs ciliary beating and sperm flagellar motility, leading to disorders such as primary ciliary dyskinesia, which manifests as chronic respiratory infections and laterality defects, and can also disrupt cerebrospinal fluid flow, causing hydrocephalus and male infertility such as asthenozoospermia. In primary cilia, reduced glutamylation perturbs intraflagellar transport and ciliary signaling and contributes to ciliopathies including Joubert syndrome and retinal degeneration. In dividing cells, altered glutamylation on centrosomes leads to errors in chromosome segregation and is associated with cancer progression. This review summarizes current knowledge of the enzymes, structural principles, and cellular mechanisms governing tubulin glutamylation, highlights its emerging roles in human diseases, and discusses new technological advances—including biochemical reconstitution, super-resolution imaging, and live-cell manipulation tools—that are beginning to reveal how this modification dynamically controls microtubule properties and the functions of flagella, cilia, and centrosomes in health and disease.
